Specimen Error Study On-Demand Webinar
Examining the Quality, Patient Safety, & Financial Impacts of Specimen Errors: See the results of the study, improvements the hospitals made, and the avoidable costs.
⏰ 4 min read
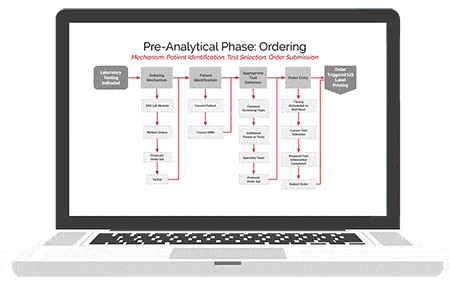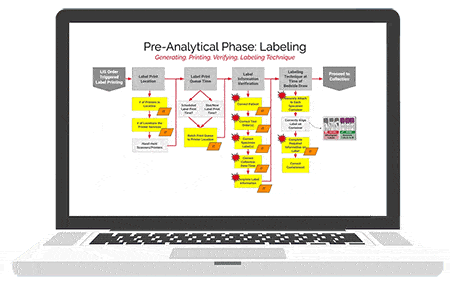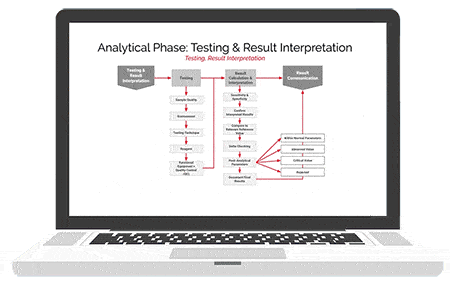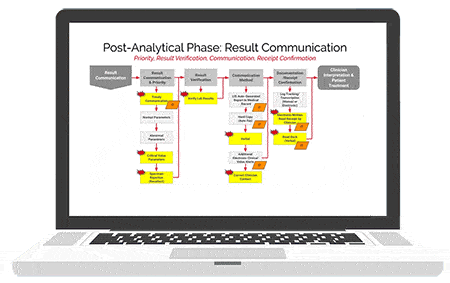 American Data Network Patient Safety Organization (ADNPSO) analyzed 4 years of data and found specimen events consistently ranked among its highest reported errors with 73.7% of the incidents deemed preventable. This led ADNPSO to develop and launch a 9-month Specimen Focused Study aimed at better understanding why these events happen and how to reduce errors across all stages of the Specimen Process (Pre-analytical, Analytical and Post-analytical).
American Data Network Patient Safety Organization (ADNPSO) analyzed 4 years of data and found specimen events consistently ranked among its highest reported errors with 73.7% of the incidents deemed preventable. This led ADNPSO to develop and launch a 9-month Specimen Focused Study aimed at better understanding why these events happen and how to reduce errors across all stages of the Specimen Process (Pre-analytical, Analytical and Post-analytical).
ADNPSO collaborated with Patient Safety and Laboratory experts to create a specimen data collection form and recruited PSO members to join the study. In partnership with the 15 acute care participants, ADNPSO performed aggregate analytics, hosted learning sessions to discuss findings, guided Rapid Cycle PDSAs, and developed detailed Process/Cause Mappings to galvanize improvement activities.
Among other successes, ADNPSO participants realized a 147% increase in specimen event reporting during the study and potentially estimated avoidable costs of $420K – $1.04M in just 9 months.
In this webinar, we’ll discuss:

 How to Make the Business Case for Patient Safety – Convincing Leadership...
How to Make the Business Case for Patient Safety – Convincing Leadership...